ChemiRise
We have developed an end-to-end, retrosynthesis system, named ChemiRise, that can propose complete retrosynthesis routes for organic compounds rapidly and reliably. The system was trained on a processed patent database of over 3 million organic reactions. Experimental reactions were atom-mapped, clustered, and extracted into reaction templates. We then trained a graph convolutional neural network-based one-step reaction proposer using template embeddings and developed a guiding algorithm on the directed acyclic graph (DAG) of chemical compounds to find the best candidate to explore. The atom-mapping algorithm and the one-step reaction proposer were benchmarked against previous studies and showed better results. The final product was demonstrated by retrosynthesis routes reviewed and rated by human experts, showing satisfying functionality and a potential productivity boost in real-life use cases.
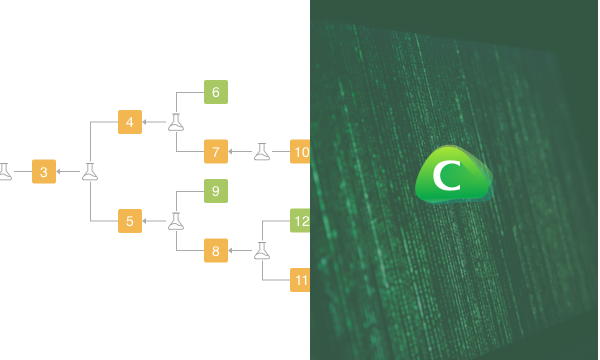

System architecture of ChemiRise, information flow from bottom to the top

Evaluation results of atom mapping algorithms
| Algorithm | Complete mapping rate | Average number of C-C bond broken |
|---|---|---|
| ChemiRise | 73% | 0.28 |
| Marvin | 63% | 0.30 |
| Indigo | 57% | 0.37 |
Evaluation results of reaction proposers
| Model | Top-N Accuracy (%) | |||||
|---|---|---|---|---|---|---|
| 1 | 3 | 5 | 10 | 20 | 50 | |
| ChemiRise | 43.8 | 62.1 | 70.1 | 78.3 | 85.0 | 90.5 |
| Coley et al. | 37.3 | 54.7 | 63.3 | 74.1 | 82.0 | 85.3 |

